 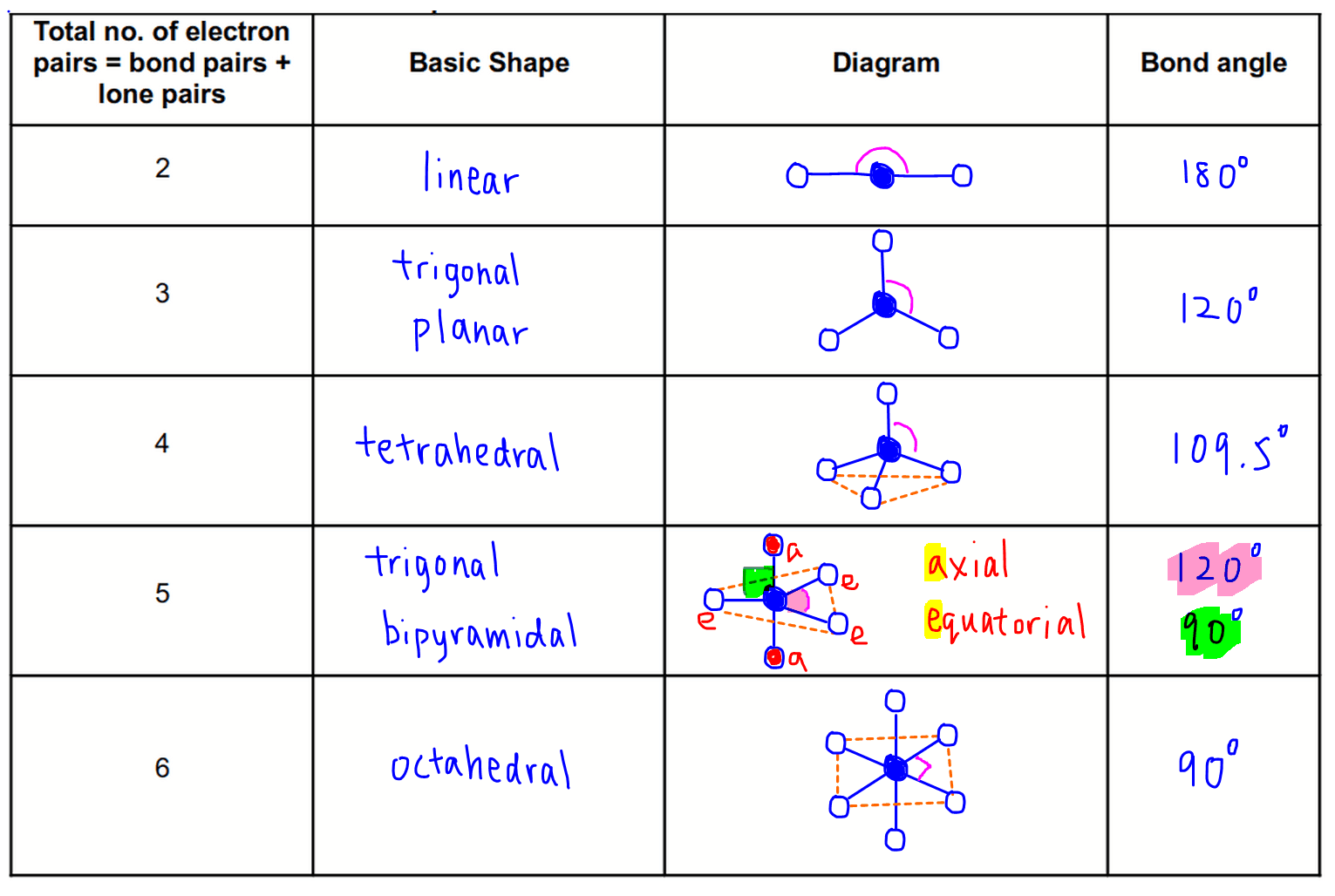 
The covalent model of chemical bonding assumes that the electron pairs responsible for bonding are concentrated into the region of apace between the bonded atoms. Like all electrons, these occupy regions of space which we can visualize as electron clouds – regions of negative electric charge, also known as orbitals – whose precise character can be left to more detailed The valence shell electron pair repulsion (VSEPR) model that we describe here focuses on the bonding and nonbonding electron pairs present in the outermost ( valence) shell of an atom that connects with two or more otherĪtoms. Is capable of rationalizing and predicting the geometry of the bonds around a given atom in a wide variety of situations.Įlectron-pairs repel each other and try to stay as far apart as possible. Nevertheless, a slight extension of the simple shared-electron pair concept The Lewis electron-dot structures you have learned to draw have no geometrical significance other than depicting the order in which the various atoms are connected to one another. Being able to understand and predict coordination geometry is an important part of chemistry and is the subject of this The local ( coordination) geometry surrounding a given atom that is covalently bound to its neighbors is constant. In most molecules, those parts joined by single bonds can rotate with respect to each other, giving rise to many different geometric forms. Geometrical shape the icosahedral C 60 soccer ball is a rare exception. In general, only the smallest molecules can be said to have a fixed Molecules are three-dimensional objects that occupy a three-dimensional world it is easy to forget this after seeing so many depictions of molecular structures on a two-dimensional page. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
